Polyols are one of the two essential building blocks in the production of polyurethane. These versatile chemical compounds contain two or more hydroxyl (–OH) functional groups per molecule, enabling them to react with isocyanates to form polyurethane structures. From the rigid insulation panels in buildings to the flexible foam in furniture, polyols determine the core properties of the final polyurethane product.
As a leading polyurethane raw material manufacturer, Kimpur produces a wide range of polyol-based systems tailored for diverse industrial applications. Understanding polyol chemistry is essential for selecting the right polyurethane system for any project.
What Is a Polyol? Definition and Chemical Structure
A polyol is an organic compound that contains multiple hydroxyl (–OH) groups. The general chemical formula for polyols used in polyurethane production can be represented as HO–[R–O]ₙ–H, where R represents the repeating unit of the polymer chain. The term “polyol” literally means “many alcohols,” referring to the multiple –OH groups present in the molecule.
In polyurethane chemistry, polyols react with isocyanates (compounds containing –NCO groups) through a condensation reaction known as polyaddition. This reaction forms urethane linkages (–NH–CO–O–), creating the polymer chain that gives polyurethane its name and properties.

The key characteristics that define a polyol include:
- Hydroxyl number (OH value): Measured in mg KOH/g, this indicates the number of reactive –OH groups available. Higher OH values generally lead to more rigid products.
- Functionality: The average number of –OH groups per molecule. Difunctional polyols (f=2) produce linear, flexible chains, while higher functionality (f=3 to 8) creates cross-linked, rigid networks.
- Molecular weight: Ranges from 200 to 10,000 g/mol. Low molecular weight polyols produce rigid foams, while high molecular weight polyols yield flexible foams.
- Viscosity: Affects processing and mixing behavior in polyurethane systems.
- Acid value: Indicates the degree of residual acidity, particularly important in polyester polyols.
Types of Polyols Used in Polyurethane Production
Polyols are classified into several categories based on their chemical backbone. Each type imparts distinct properties to the resulting polyurethane product. The two dominant categories—polyether polyols and polyester polyols—account for the vast majority of global consumption, with polyether polyols representing approximately 80–90% of total usage.
Polyether Polyols
Polyether polyols are hydroxyl-functional oligomers produced by the ring-opening polymerization of cyclic ethers, primarily propylene oxide (PO) and ethylene oxide (EO). These polyols feature ether bonds (C–O–C) along their backbone chain.

The production process involves propoxylation or ethoxylation of starter molecules (such as glycerol, trimethylolpropane, or sucrose) in the presence of catalysts. By varying the ratio of propylene oxide to ethylene oxide and the choice of starter molecule, manufacturers can precisely control the polyol’s molecular weight, functionality, and reactivity.
Key properties of polyether polyols:
- Excellent hydrolytic stability — resistant to moisture degradation
- Good low-temperature flexibility
- Lower viscosity, making them easier to process
- Cost-effective for large-scale production
- Widely used in flexible foams, rigid foams, and elastomers
Polyether polyols are the preferred choice for applications where moisture resistance and flexibility are critical, including furniture cushioning, automotive seating, mattress foams, and spray polyurethane foam insulation.


Polyester Polyols
Polyester polyols are produced through the polycondensation reaction of dicarboxylic acids (or their anhydrides) with glycols. The ester bonds (–CO–O–) in their backbone give these polyols a distinct set of properties.

Common dicarboxylic acids used include adipic acid, phthalic acid, isophthalic acid, terephthalic acid, and maleic anhydride. Glycol components typically include ethylene glycol, diethylene glycol, propylene glycol, 1,4-butanediol, and 1,6-hexanediol. The specific combination of acid and glycol determines the final properties of the polyester polyol.
Polyester polyols are divided into three subgroups:
- Aliphatic polyesters: Based on linear diacids like adipic acid and succinic acid. They offer good mechanical properties and flexibility.
- Aromatic polyesters: Based on cyclic diacids like terephthalic acid and isophthalic acid. They have lower molecular weights but provide superior heat and fire resistance.
- Caprolactone-based polyesters: Produced by ring-opening polymerization of caprolactone. They provide excellent hydrolysis resistance among polyester types.
Key properties of polyester polyols:
- Superior mechanical strength and abrasion resistance
- Better heat and fire resistance compared to polyether polyols
- Excellent solvent resistance
- Strong crystalline structure within urethane linkages
- Disadvantage: susceptible to hydrolysis under high humidity and temperature
The branching and molecular weight of polyester polyols directly determine the type of polyurethane produced. Highly branched, low molecular weight polyester polyols yield rigid polyurethane foam with excellent thermal and chemical resistance. Conversely, less branched, high molecular weight polyester polyols produce flexible polyurethane foams with high elasticity.

Polycarbonate Polyols
Polycarbonate polyols contain carbonate linkages (–O–CO–O–) in their backbone. They are produced by the reaction of diols with phosgene derivatives or through transesterification with dialkyl carbonates.
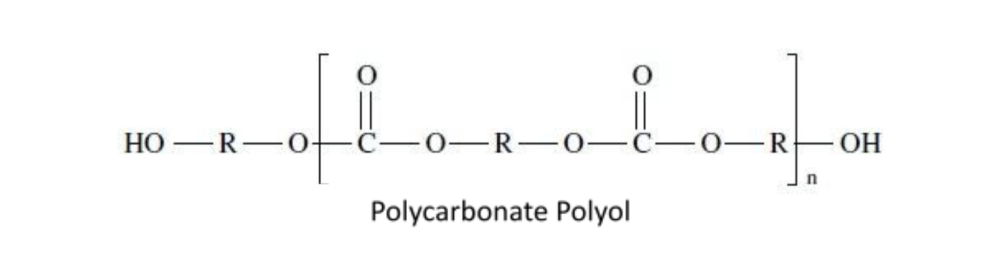
These polyols offer an exceptional combination of properties: excellent hydrolysis resistance (superior to standard polyester polyols), outstanding weathering stability, and high mechanical strength. They are preferred for high-performance polyurethane coatings, premium elastomers, and medical-grade polyurethanes where long-term durability is paramount.
Bio-Based and Recycled Polyols
Sustainability is driving the development of bio-based polyols derived from renewable feedstocks such as castor oil, soybean oil, rapeseed oil, and palm oil. Castor oil is particularly valuable because it naturally contains hydroxyl groups, making it directly usable as a polyol without extensive chemical modification.
Recycled polyols, produced through the glycolysis of post-consumer polyurethane waste, represent another important category. These polyols contribute to the circular economy by converting end-of-life polyurethane products back into usable raw materials. Kimpur’s sustainability approach includes developing green polyol-based polyurethane systems that reduce environmental impact while maintaining product performance.
Polyol Properties and Their Effect on Polyurethane
The relationship between polyol properties and polyurethane performance is direct and predictable. Understanding this relationship is essential for formulators and engineers selecting the right system.
| Polyol Property | Low Value → Result | High Value → Result |
|---|---|---|
| Molecular Weight | Rigid foam, hard coatings | Flexible foam, soft elastomers |
| Functionality (f) | Linear chains, thermoplastics | Cross-linked networks, thermosets |
| OH Number | Flexible products (28–160 mg KOH/g) | Rigid products (250–800 mg KOH/g) |
| Viscosity | Easier processing, faster mixing | Slower processing, may need heating |
Industrial Applications of Polyols
Polyols serve as the foundation for polyurethane systems across virtually every major industry. Their versatility allows manufacturers to engineer products ranging from ultra-soft comfort foams to structural components that bear heavy loads.
Construction and Insulation
In the building sector, polyols are formulated into rigid polyurethane and polyisocyanurate (PIR) foam systems that provide the highest thermal insulation efficiency of any commercially available material. These systems are used in sandwich panels, pipe insulation, roof and wall insulation, and spray foam applications. The low thermal conductivity (λ = 0.020–0.028 W/mK) of polyurethane rigid foam makes it significantly more efficient than traditional insulation materials.
Furniture and Comfort
Flexible polyurethane foam, produced from high molecular weight polyether polyols, is the dominant cushioning material in furniture, mattresses, and automotive seating. Viscoelastic (memory) foam, a specialty flexible foam, uses specially designed polyol blends to achieve its signature pressure-relieving properties.
Footwear
The footwear industry relies heavily on polyol-based polyurethane systems for shoe soles, insoles, and midsoles. Polyurethane soles offer an unmatched combination of durability, comfort, lightweight properties, and design flexibility compared to rubber or EVA alternatives.
Coatings, Adhesives, Sealants, and Elastomers (CASE)
In CASE applications, polyols enable the formulation of high-performance polyurethane coatings for floor protection, automotive finishes, and industrial surfaces. They are also the basis for structural adhesives, weatherproof sealants, and durable elastomers used in wheels, rollers, and vibration dampening components.
Automotive
The automotive industry uses polyol-based polyurethane systems in seat cushions, headrests, armrests, steering wheels (integral skin foam), sound insulation, and lightweight structural components. Polyurethane’s ability to replace heavier materials helps automotive manufacturers reduce vehicle weight and improve fuel efficiency.
How Kimpur Produces Polyol-Based Polyurethane Systems
As a vertically integrated polyurethane raw materials manufacturer, Kimpur produces polyester polyols and formulates complete polyurethane systems at its state-of-the-art production facilities. By controlling the entire production chain from raw material synthesis to final system formulation, Kimpur ensures consistent quality and the ability to develop customized solutions for specific customer requirements.
Kimpur’s polyol-based product portfolio includes systems for rigid foam insulation, flexible foam, integral skin foam, spray foam, coatings, and adhesives — serving markets across Turkey, Europe, and globally through the Kimpur Europe network.
Frequently Asked Questions About Polyols
What is the difference between polyester polyol and polyether polyol?
Polyester polyols offer superior mechanical strength, abrasion resistance, and heat resistance, making them ideal for coatings, elastomers, and rigid foams. Polyether polyols provide better hydrolytic stability, lower viscosity, and greater flexibility, making them the preferred choice for flexible foams, spray foams, and applications exposed to moisture. Approximately 80–90% of polyols used globally are polyether polyols.
What is polyol used for?
Polyol is one of the two main components (alongside isocyanate) used to produce polyurethane. Polyurethane products made from polyols include building insulation panels, spray foam, furniture cushions, mattresses, shoe soles, automotive parts, coatings, adhesives, sealants, and elastomers. The type and properties of the polyol determine the characteristics of the final product.
How is polyol produced?
Polyether polyols are produced by ring-opening polymerization of propylene oxide and/or ethylene oxide onto starter molecules (such as glycerol or sorbitol) using catalysts. Polyester polyols are produced by polycondensation of dicarboxylic acids with glycols. Bio-based polyols can be derived from natural oils like castor oil through chemical modification.
What is the role of polyol in polyurethane foam?
Polyol forms the “soft segment” of the polyurethane polymer chain, providing flexibility, elasticity, and resilience. The molecular weight, functionality, and chemical type of the polyol directly determine whether the resulting foam is rigid or flexible, its density, compression strength, thermal properties, and durability.
Are polyols toxic or hazardous?
Polyols are generally considered to have low toxicity compared to many industrial chemicals. They are not classified as hazardous materials for transportation purposes. However, as with all industrial chemicals, proper handling procedures, personal protective equipment, and safety data sheet guidelines should be followed during storage and processing.







